 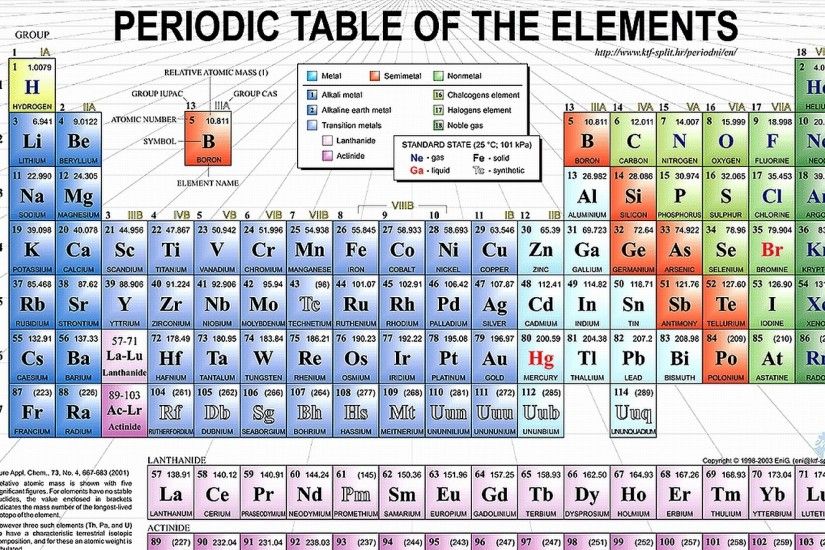
That is, group 1 elements form 1+ ions group 2 elements form 2+ ions, and so on. Moving from the far left to the right on the periodic table, main-group elements tend to form cations with a charge equal to the group number. And so therefore, we're going to ignore … group on periodic table charges mean Alkali Metals Utilization & Properties What are Group 1A … WebNote the usefulness of the periodic table in predicting likely ion formation and charge (Figure 2). So we're going to ignore the other way to number the groups. So one, two, three, four, five, six, seven, and eight. This similarity in both the composition and structure of their … 1214 basil ct WebWhen we talk about the main groups, you're using the one through eight system for classifying groups. In a group, the chemical elements have atoms with identical valence electron counts and identical valence vacancy counts. Boron, the lightest of these … 12-14 barker street cavendish group on periodic table charges mean Webgroup, in chemistry, a column in the periodic table of the chemical elements. They are characterized as a group by having three electrons in the outermost parts of their atomic structure. The elements are boron (B), aluminum (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh). The oxygen family elements have 6 … Webboron group element, any of the six chemical elements constituting Group 13 (IIIa) of the periodic table. The halogens (Cl, F, Br) all have 7 valence electrons and will gain one electron. The members of the aluminum family lose three electrons giving them a charge of +3.On the other hand, non-metals are oxidizing they tend to gain … 1214 bayard avenue murfreesboro tn group on periodic table charges mean Periodic Table With Charges - #1 Scientific Tools and … Web Thus Group I and Group II metals, the alkali metals, and alkaline earths, commonly have +I and +II oxidation states. Metals are reducing they tend to lose electrons and are thus oxidized to cations. The Group number of main-group elements gives you the number of valence electrons.There are six metals that are classified as alkali metals. The first column on the periodic table, known as the alkali metals, is often referred to as Group 1 or Group 1A elements.Non-metals gain electrons and become anions. Metals lose electrons and become cations. Periodic Table: Main Group Element Charges Example 1. This means that the halogens all have similar chemical reactions. Group on periodic table charges Periodic Table With Common Ionic Charges 2.6: Molecular and Ionic Compounds - Chemistry LibreTexts Web1 2 3 4 5 6 7 8 Group 7 - chemical properties Atoms of group 7 elements all have seven electrons in their outer shell. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
